 Facebook
Facebook
 X
X
 Instagram
Instagram
 TikTok
TikTok
 Youtube
Youtube
It reads like a bad science fiction novel: A small Illinois biotech company cuts a deal with UCSD. The university agrees to test a substitute for human blood on comatose patients — victims of gunshots and car crashes — without the patients' consent. Within the city of San Diego, the experiment is targeted at several neighborhoods south of I-8, where many poor and minority residents are unlikely to have heard of the study and unlikelier still to have the resources to sue if something goes awry. The university conceals the identity of the city's paramedic units who carry the blood substitute.

When a curious reporter asks for the names of the neighborhoods where the study is being carried out, a research coordinator working for the university tells him that the Chicago sponsor doesn't want the information made public because Wall Street moneymen — hoping to reap a financial windfall from their investment in the company — might sell if they discover the experiment is not going well. When the reporter asks university officials to provide details of two "adverse event" reports involving the study, the officials stonewall, saying the information is "nonpublic" because the university has signed a confidentiality agreement with the laboratory. But if Hollywood wouldn't buy that scenario, the University of California and the City of San Diego already have. A little more than two years ago, officials at UCSD signed an agreement with Northfield Laboratories for clinical trials of PolyHeme.

Despite repeated studies, PolyHeme has yet to be approved by the Food and Drug Administration, and this May, Forbes magazine told its predominantly Wall Street audience that researchers at Walter Reed Army Institute of Research had "published a study suggesting PolyHeme might not be so safe after all. Comparing resuscitative fluids, it found that hemorrhaging lab rats treated with PolyHeme experienced higher mortality rates than those that received two commonly used solutions."
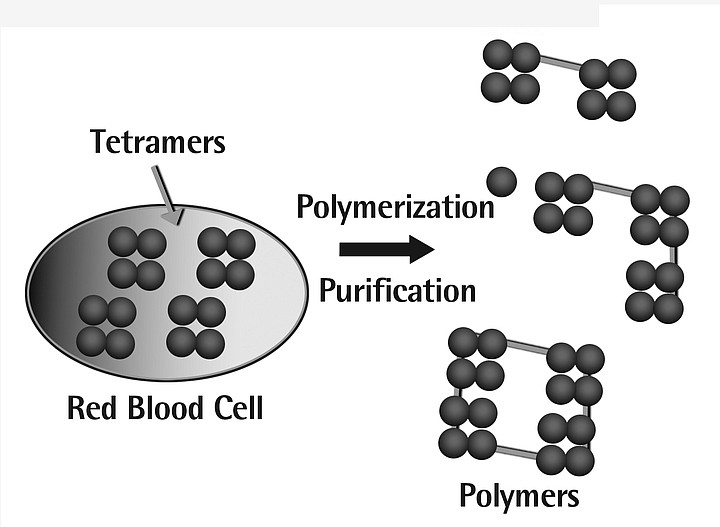
First, hemoglobin is extracted from red blood cells and filtered to remove impurities. This purified hemoglobin is then chemically modified and purifies using a multi-step process to create a polymerized form of hemoglobin. The modified hemoglobin is then incorporated into an electrolyte solution.
Polymerization is intended to eliminate the undesirable effects historically associated with hemoglobin-based blood substitutes: vasoconstriction, kidney dysfunction, liver dysfunction, and gastrointestinal distress.
The founder of Northfield labs is Steven Gould, a 57-year-old physician from Chicago who has worked on blood substitutes for more than 25 years. In 1979, after obtaining a grant from the Army, he and several other doctors at the University of Chicago developed a technique to separate hemoglobin, the oxygen-carrying protein, from blood, then link the hemoglobin molecules together into polymers, hence the name PolyHeme.
"Initially a project of the United States Army following the Vietnam War," Northfield's website says, "the objective was to develop a preparation that could be infused rapidly and in massive quantities to injured battlefield combatants in far-forward areas and mobile surgical theaters before the injured could be evacuated to more sophisticated field hospitals for definitive care."
Northfield's promotional material describes PolyHeme as "a solution of chemically modified human hemoglobin, which simultaneously restores lost blood volume and hemoglobin levels and can be rapidly and massively infused." The advantages of PolyHeme are that it "requires no cross-matching. It is compatible with all blood types and therefore immediately available for infusion. It has an extended shelf life of over 12 months."
The Army continued to fund Gould until 1984, when it began supporting his larger rival, Baxter International, in its own quest for a blood substitute. At that point, Gould shifted his aim and put together $1 million in funding from private investors. A decade later, in 1994, with several small clinical trials under his belt, Gould took Northfield public, raising $15 million at $6.50 a share.
But in 2001, according to the Forbes article, the Food and Drug Administration rejected Northfield's application to approve PolyHeme for general use, citing lack of sufficient clinical studies. The stock tanked, sinking from $13.25 to $3.05.
Gould held on, sacking the company's chief executive and eventually reaching an agreement in 2003 with the FDA to allow a new kind of clinical trial that could be run without getting the patient's consent. Employing a little-used exemption in FDA regulations, Gould proposed having paramedics in cities across America infuse PolyHeme into 360 patients. San Diego was one of those cities.
Under Northfield's deal with the FDA, the company and its research partners agreed to conduct public information campaigns in each city where PolyHeme was to be deployed. They were to arrange newspaper, radio, and television coverage advising the public of the tests and of the opportunity to opt out by wearing a special bracelet. They were to set up a website containing voluminous information about the nature of the study and the right to opt out. In San Diego, much of this didn't come to pass.
Notice of the trial was posted on an obscure part of a website run by the UCSD Human Research Protections Program. Today, those who click on a link labeled "Public Notices" are taken to another link labeled "PolyHeme Trauma Trial" and thence to the study's information page. Readers are told that "This groundbreaking study is being conducted in accordance with federal regulations permitting an exception from informed consent requirements under 21 CFR 50.24." The statement says that a public information meeting, where "comments and concerns" were to be solicited, would be held August 6, 2003, at the UCSD Medical Center in Hillcrest. "Members of the media are invited to cover this event," it added.
The only major newspaper story covering the trial was spun heavily in favor of the wonders of the experimental substance and made light of its possible hazards. "A new hope for saving of lives," said the headline over the March 23, 2004, story in the Union-Tribune. "Paramedics to conduct test of blood substitute."
"These are people who are facing death in a few hours," the story quoted Dr. David Hoyt, trauma director at UCSD Medical Center and principal investigator of the local PolyHeme study. "Giving them this blood substitute might just save them when conventional treatment wouldn't." The paper added that "In a study of PolyHeme from the late 1990s, 75 percent of those treated with it survived, compared with 30 percent treated only with saline solution."
The paper went on to report that "the blood substitute will only be available to paramedics working south of Interstate 8." Specific paramedic and ambulance units carrying PolyHeme were not identified. "Designated paramedic units in San Diego and the South County and the Mercy Air helicopter crew will have a cooler containing bags of the substitute," the story added.
The clinical trial began in San Diego County on May 20, 2004, when a Mercy Air emergency-services helicopter, based at Gillespie Field, started carrying PolyHeme. On June 11, 2004, seven ground ambulances serving cities outside of the city of San Diego joined the trial. The ground ambulances are owned by a private company, American Medical Response, which serves 11 cities in the county as well as unincorporated areas. According to the company's spokesman, Devon Price, the ambulances participating in the study operate in National City, Chula Vista, Bonita, and Imperial Beach. Earlier this week, Hoyt, of UCSD, said six paramedic rigs outside the city of San Diego were equipped with PolyHeme.
But according to records obtained from the City of San Diego, at the time the U-T story appeared, city officials and UCSD administrators were still negotiating when and where the city's paramedics would use PolyHeme.
Northfield wanted to limit the PolyHeme trial to neighborhoods with the highest trauma rates. On May 2, 2003, Beth Romeril, the research coordinator of the UCSD study, wrote Hoyt, the principal investigator. "My biggest concern at this time is that in conversation with Bob McGinnis from Northfield on Wednesday, he indicated to me that Northfield would not have enough product to stock even the 12 or so ambulances in UCSD's catchment area, that part of the reason for the TREG [San Diego County Regional Trauma System Registry] search would be to identify the '3 or 4' ambulances that would be most likely to encounter appropriate patients.
"I have asked him to confirm the maximum number of ambulances they would be able to supply for us and waiting to hear back," Romeril wrote, adding that her geographic survey of trauma data was complicated. "Most of the time the location (zip code) where the encounter took place is not available, so we can't identify specific medic rigs in most cases."
At the City of San Diego, officials worried about possible lawsuits in case PolyHeme didn't work as advertised. They wanted to isolate the city's possible financial liability by demanding that the study's sponsor indemnify the city for $15 million against malpractice claims.
Why might there be malpractice claims?
By the very definition of the study, all patients are in hemorrhagic shock and unable to refuse the PolyHeme. As noted by Northfield: "It is expected that patients enrolled in this trial will be unable to provide informed consent because of the nature and extent of their injuries." The lack of consent was justified on the grounds that the existing blood substitute currently used by paramedics is simple saline solution and that almost anything, even an experimental substance like PolyHeme, would be an improvement for desperate trauma patients.
And there was another reason for the city to fear lawsuits. Even after arriving at the hospital, patients would not receive real blood.
The protocol called for trauma patients selected for the study to be randomly separated into two groups: the control group would get the standard saline as the resuscitative fluid; the experimental group would be infused with PolyHeme. When patients arrived at the hospital, those receiving saline would be switched to blood. But those receiving PolyHeme would continue to receive PolyHeme -- up to a maximum of six units -- for 12 hours after their injury.
Other medical centers were not as sanguine as UCSD about the PolyHeme trial and pulled out before it began. According to a letter from Northfield to Hoyt, dated last August 27, Milwaukee's Froedtert Memorial Lutheran Hospital's review board "has not approved conduct of the study at their institution. The letter from [redacted] cites this [review board's] position that once the patient is in the hospital, blood is not an unsatisfactory treatment, which is what is required for waived consent."
Boston Medical Center dropped out for similar reasons.
A study at the University of Southern California was shut down before it began, according to state records.
The City of San Diego moved forward with the trial in midsummer. In a memo dated August 10, 2004, the city's Emergency Medical Program manager, Donna Goldsmith, quietly informed the council that the PolyHeme trial would finally proceed. "Based upon previous trauma demographic data," the memo said, "it is projected that the San Diego region will contribute 40 patients to the study. The safety and efficacy of this artificial blood product have been demonstrated in several in-hospital studies involving more than 300 patients."
Goldsmith's memo went on to say that paramedic units in three of the city's poorest communities -- Oak Park, Nestor, and San Ysidro -- had been chosen for the trial because those areas generate the largest number of severe trauma patients, who typically are victims of car accidents and shootings. (A fourth paramedic unit, based downtown, would later be included in the study, Goldsmith said in a recent telephone interview, but that was not included in the memo to the council.)
"Specific ambulances and medic-engine crews have been selected to carry PolyHeme in specially designed coolers," the memo said. "The crews selected are those that frequently encounter major trauma victims within the catchment regions of the trauma centers participating in the trial -- UCSD Medical Center and Scripps-Mercy Hospital."
The units assigned to the PolyHeme trial -- Oak Park's Medic 26, San Ysidro's Medic 29, and Nestor's Medic 62 -- rarely venture from their home turf, Goldsmith said.
"Although PolyHeme will be placed on the engines and ambulances listed above, due to the fluidity of the EMS System, these resources could be assigned as needed throughout the City."
But under the terms of the study, paramedics could administer PolyHeme only if they were delivering the patient to the UCSD Medical Center or to Scripps Mercy Hospital.
The agreement did not go to council. It is dated September 28, 2004, and is signed by then-deputy city manager Patricia Frazier. The trial began in the city on November 19.
(In May of this year, Frazier, along with former treasurer Mary Vattimo and human resources director Cathy Lexin, quit her job in what was widely seen as a shake-up related to the city's festering pension-fund crisis. Lexin and Vattimo, former members of the pension board, were later charged with conflict of interest for their votes to allow underfunding of the pension system. Frazier was not charged.)
Though they had become part of the nationwide PolyHeme trials, residents of the neighborhoods served by the designated paramedic units were none the wiser. Many details of the PolyHeme study's protocol, along with much other information about the trial in San Diego, have never been made public. In a letter dated January 16, 2004, Gould told Hoyt, principal investigator of the UCSD study, "I urge you to remind your study coordinator and all others associated with the trial that information about patients and the details of the trial at your site are not to be shared with anyone outside the study team."
Under its August 2004 agreement with UCSD, the City of San Diego agreed "not to disclose to any third party any Confidential information," defined as "all information, data, materials, in whatever form or medium, that [Northfield] treats as confidential and proprietary information."
The secrecy has been sweeping, denying citizens any chance to monitor the study's progress or receive information about possible adverse effects on patients. A "research plan," dated January 10, 2003, and attached to the city's contract with UCSD, was obtained from the city after repeated requests under the California Public Records Act. The plan spells out details of the PolyHeme trial, but before UCSD turned it over earlier this year, it was heavily redacted to remove any hint of where and how the substance is being tested or what paramedics are to do in case of life-threatening side effects.
One section of the plan, which describes how UCSD researchers would attempt to get the consent of a comatose patient's relative before proceeding, was gutted. It begins:
"Once a patient is enrolled, the investigator will commit to diligent and frequent attempts to contact a legally authorized representative or a family member in order to provide them with the opportunity to object to the patient's continued participation in the study. At a minimum, contact with a legally authorized representative or family member by the study coordinator will be attempted at least:"
The paragraph that follows, apparently describing the mandated conditions for obtaining consent before proceeding, is completely blacked out.
Particularly troubling to some observers are two in-house university reports of "adverse events" that have occurred.
A letter dated May 27, 2004, from Daniel Masys, M.D., then director of UCSD's Human Research Protections Program, to Hoyt discussed the first incident. "Your April 29, 2004 adverse event report for Project 030443 has been reviewed and accepted by the IRB Committee at the May 20, 2004 meeting," Masys wrote. The IRB, or Institutional Review Board, is a group set up by the university to review the ethics and integrity of research activities.
The letter's next sentence, apparently describing the nature of the adverse event, was entirely redacted by the university. Two final sentences followed the redaction: "The UCSD IRB believes that all issues have been adequately addressed and feels that the decision to support this protocol in July 2003 was appropriate. Thank you for keeping us informed."
Asked how an adverse event could have occurred in April 2004, almost a month before the study began on May 20, Beth Romeril, the UCSD research coordinator affiliated with the study, denied that the May 27, 2004, letter regarding an adverse event report had anything to do with the PolyHeme study, even though the document specifically refers to the trial. Romeril added that university staff fill out a lot of forms and sometimes forget to change the heading at the top, so they might have left the wrong project number on the document. She offered no explanation as to why the letter also makes reference to the project number in its body.
Another letter from Masys to Hoyt, dated August 26, 2004, referenced a subsequent incident. "Your July 22, 2004 adverse event report for Project 030443 has been reviewed and accepted by the IRB Committee at the August 2004 meeting. This report concerns the following subject: 1)." An entirely redacted sentence, apparently describing the specifics of the adverse event, followed.
The letter then concluded: "It is your opinion that the event is definitely unrelated to the study. It is also your opinion that this update will not require a change to the risk management procedures, application, informed consent document, or the risk/benefit ratio. Thank you for keeping us informed."
When asked to provide copies of the reports referred to in the letters, the university produced a form labeled "Report of Adverse Event," signed by Hoyt, dated July 27, 2004. Two pages of the form, under the heading "Principal Investigator's Assessment of Event," were completely blank, with the exception of the word "redacted" stamped repeatedly down the empty pages. Other information, such as the date of occurrence and whether it caused a fatality or was life-threatening, was also blacked out.
In the April 28, 2005, cover letter that accompanied the material, Linda L. Maczko, the university's Public Records Act coordinator, said, "The basis for the redaction to the adverse event report and consent form is patient privacy.... The basis for all the other redactions made to the documents provided is trade secret/proprietary information."
Legal experts familiar with public records law note that patient privacy can be protected by redacting the patient's name and identifying information and thus is not grounds to withhold other details of the event.
They also maintain that in such situations where a public agency may place members of the public at risk without their consent, the state's Public Records Act requires the agency to provide the public with as much information as possible.
"We filed this records request originally with UCSD on December 23, 2004," notes William Sauer, the attorney who has handled the Reader's repeated Public Records Act requests to the university. "UCSD has continually 'shielded' the results of the trial, alleging it is a trade secret and its release violates patient confidentiality.
"We have asked for no individual patient information. We have asked for no secret formulas or processes. UCSD is a public university that should respond promptly to the CPRA. Instead it shields and protects drug companies that pay it for the clinical trials.
"UCSD should carefully review the recent overwhelming approval by the voters in California of Prop. 59, commonly referred to as the Sunshine Amendment. Frankly, we haven't seen delaying tactics like this in 25 years of legally obtaining public documents."
Under a "clinical study agreement" between UCSD and Northfield dated March 22 of last year, the university is to collect $10,840 "per Completed Patient" in the study. Forty patients were anticipated.
The contract itself -- provided by the university, but heavily redacted -- outlines a "payment schedule" based on how long a patient remains in the trial. If a patient drops out early in the course of treatment, before the goals of the timeline are met, UCSD receives significantly less money. That might create a conflict of interest, says Nancy King, a bioethicist, between the university's ethical mandate to fully inform patients and relatives about the dangers of the treatment and the university's desire to get fully paid by Northfield.
Because the contract was heavily redacted, a full understanding of the payment schedule is difficult. For example, the contract says that "for each Qualified Patient completing step 1 who withdraws from the study for any reason prior to completing [redacted]," Northfield will pay the university just 35 percent of the $10,840 fee. "For each Qualified Patient completing step 1 [redacted] phase," the fee would be 50 percent of the total, and so forth.
No matter how many patients are enrolled or how long they remain in the study, the contract says, "Non-refundable Start Up costs of $39,106.00 plus the cost of four (4) completed patients ($43,360.00 less 20% ($8,672) = $34,688) for a total of $73,794.00 will be payable upon full execution of the Agreement." All costs are subject to a 3 percent annual adjustment for inflation.
Scripps Mercy Hospital is also paid for participating in the study. According to a contract between UCSD and Scripps Health, dated May 11 of last year, UCSD sought to "conduct part of this trial" at Scripps Mercy "in order to enroll a larger number of patients." Using funds provided by Northfield, the contract says, UCSD agreed to pay Scripps Mercy Hospital a $2000 fee for each trauma patient who completed the PolyHeme trial, as set out in its agreement with Northfield. Of that $2000, the hospital receives $878 for lab fees and Dr. Michael Sise, head of trauma care at Scripps Mercy, collects $1122 per patient, the contract says.
According to an e-mail from Scripps Health public relations director Don Stanziano, the payment to Sise is not for his sole benefit. "While reimbursement is sent to Dr. Sise -- lead investigator at Scripps Mercy, he (the Scripps Mercy Trauma Department) distributes the monies to cover the costs as outlined in the study's approved budget.
"Physicians receive reimbursement for the extra time it takes to attend to the study. The trauma surgeons (four at Scripps Mercy) receive $560 per patient. [That] money is donated to Scripps Mercy's research fund to offset the cost of doing unfunded clinical research. The remaining funds pay for other staffing costs such as nursing."
Stanziano declined to comment on whether Mercy had suffered any adverse events related to the study. "Due to confidentiality of study protocols, that question must be directed to Northfield Labs, the study's sponsor," the e-mail said.
Under state law, the medical director of the county's Emergency Medical Services, Dr. Gary Vilke, is supposed to act as watchdog over all paramedics in San Diego County. But that is not Vilke's only job. The rest of his time is spent as medical director of prehospital services at UCSD Medical Center, where Hoyt is a member of the board of governors.
In the fall of 2003, Vilke sought permission from Richard E. Watson, the interim director of the state's Emergency Medical Services Authority, to begin testing PolyHeme in San Diego. "Initially I had concerns about the study due to the exception for patient consent," Watson responded to Vilke on October 23, 2003.
"My approval, however, is based on your agreement to do the following. For the first two months following initiation of the trial study, I would like a weekly update on the study via a phone call to me or Dr. Tharratt. For the duration of the study would also require immediate notification (by phone or fax) to Dr. Tharratt of any occurrence of a serious adverse event...and a written trial study progress report every 6 months."
Vilke reported to Watson on the study's progress in a letter dated last December 16. Despite the two adverse event reports on file at UCSD, Vilke reported that "There were no complications in the study protocol, nor were there any complications or adverse events in the care of the patients in pre-hospital or hospital setting."
Dr. Steven Tharratt, who is a medical consultant to the state's Emergency Medical Services Authority, says the authority has not been provided copies of the two adverse event reports filed by Hoyt. Despite Watson's requirement that Tharratt be notified immediately of a serious adverse event, Tharratt said the adverse event reports sent to the IRB "wouldn't be something we would be interested in seeing. That is not our area of oversight."
Vilke also reported in his letter to Tharratt that as of last December, the UCSD study had enrolled only four patients. Vilke added, "All 4 patients enrolled in the study are doing well."
Six months later, after he had been queried by a reporter about the adverse event reports, Vilke reported to Watson, in a letter dated June 20, 2005, that "to date, we have enrolled a total of fourteen patients into the study.
"There have been a total of four protocol violations. All have been reviewed by me and none resulted in any adverse events with the patients. There were no complications as a result of the study protocol, nor were there any complications or adverse events in the care of the patients in pre-hospital setting.
"We have not had any immediately reportable safety events, so no IND safety reports have been generated to date. All patients enrolled have done well and are all alive at 30-day follow up.
"We have monitored the scene times for these patients and they were essentially unchanged compared with similar status trauma patients during the same time period."
According to Vilke's letter, at least 250 patients from across the nation have been enrolled so far in the study.
But finding suitable test subjects has not been easy. Struggling to recruit subjects to the PolyHeme trials, researchers in December considered offering paramedics "incentives" in return for their cooperation, according to documents released by UCSD after more than five months of delay.
"Are there any incentives/activities that would motivate the EMS that you use or would recommend using that would not be inappropriate or be equated to directly rewarding them for patient recruitment?" asks Northfield in an agenda, dated December 8, 2004, for an "Enrollment Meeting" at its Evanston, Illinois, headquarters. The apparent response from UCSD, highlighted in boldface type: "Gifts of food items (Christmas baskets: coffee) or inexpensive give-aways like pens, penlights, mugs with 'Polyheme study' on them for the whole group regardless of enrollment as a thank-you for participation."
The agenda asks, "Do you know of any incentive program restrictions that might exist at your hospital?" The response: "No monetary gifts or payment for enrollment."
The document also said, "Share with us the worst comment you've heard from EMS/Flight Nurses (or any other study staff) regarding the study. What would be a creative response?"
The reply: "Flight crew from North County: We bring you maybe one trauma patient/year that would qualify for this study, so why did they bother including us?
"We have responded that there are so few patients that meet criteria, that every single one is precious for the study, plus the fact that the air crews are potentially the most reliable at following study protocols and thus providing the best data."
One of the most outspoken critics of the PolyHeme study is Nancy King, a professor of social medicine at the University of North Carolina at Chapel Hill School of Medicine. Trained as a lawyer and specializing in bioethics and health law, King has concentrated on issues of informed consent, a specialty that ultimately led her to monitor closely the genesis of the PolyHeme study.
"When one gets involved in a research study," King says, "normally one goes through a fairly extensive consent process and signs a consent form and is given a lot more information about what's going to happen than may well happen in the doctor's office when the doctor says, 'I think you need such and such.' "
That started to change back in 1996, King says, when Baxter International, the large pharmaceutical company, petitioned the federal government to allow testing of its blood substitute on patients who could not provide their consent.
"That's a very big departure from all of the regulatory structures that govern human-subjects research. What was ultimately approved was a regulation that permitted research under those limited circumstances where the subject is in a life-threatening condition.
"One of the things that has been quite interesting to follow since that regulatory structure was put in place was...well, there haven't been that many studies done under the emergency waiver. And people have a lot of questions about how to do it well."
Is the waiver of informed consent necessary to conduct medical research? King says there may be circumstances that make it impossible to require patient consent before an experimental treatment is administered, but they are few and far between.
"I think that most of the time, and frankly almost all of the time, there are ways to do relevant studies where the waiver is not needed. What the waiver gets you really is faster enrollment of subjects.
"You may have a scientific reason for saying we need to get more subjects quickly, but more often it's a question of how fast a drug or device or procedure that's new needs to move through the system in order to be most profitable for the study sponsor.
"One of the concerns that I have about the PolyHeme trial is that, looking at the study sponsor's website, their materials basically talk about the future of PolyHeme as being in military applications and for elective surgery. In both of those populations one can get prospective consent. So the very design of the PolyHeme study is troubling since there are other ways to gather the data.
"The other thing that has troubled me -- and this, I think, is a flaw in the IRB process as well as in the way the sponsor's thinking about the study -- is that the kinds of things that are being done per community consultation and the material that's available for community consultation are very slim.
"Most sites have merely posted notices about public meetings held in places that people don't usually go, and the investigator will go and give the slide show that's given to him by the study sponsor and hand out materials that have been developed by the study sponsor, which is all the same stuff [at every site].
"And it's all fairly flawed, because the basic materials, the basic FAQ sheets about the study, talk about how many 'patients' have already been 'treated' with PolyHeme, and that's a very misleading description.
"Nobody who has gotten PolyHeme has been anything but a research subject, because this is an experimental intervention. So anybody who gets it is a research subject. And it's not a treatment yet because it hasn't been approved. So saying, 'Well, X number of patients have already been treated with PolyHeme' provides a false sense of reassurance to a subject that may actually be quite misleading."
Northfield has carefully guarded its research protocol, the plan detailing how its study is to be conducted, the dosage to be used, the results of prior testing, and other details. King argues that this is common in the drug industry but not appropriate for research on subjects who do not provide their consent.
"What was shown in earlier-phase research? What kind of earlier-phase research was actually conducted? What numbers are there? What leads up to saying this is the right time for this kind of study now?"
Without such information, King says, communities can't judge the trade-offs between PolyHeme and saline solution, the currently used alternative.
"What ambulances can do is give you saline to keep up your blood volume, but they can't actually provide you with blood that is able to enable your system to keep getting enough oxygen," she explains. "They've got to give you saline and try to stop the bleeding and get you to the hospital, where you can get blood, because that's what carries oxygen.
"In the field then, if the argument is that PolyHeme can supply some of that oxygen at least temporarily, and it doesn't need to be refrigerated, and it doesn't need to be typed and cross-matched, well, that would be a really good thing to have."
But once a patient gets to the hospital, where blood is available for transfusion, the equation changes. PolyHeme-study patients are forbidden to immediately receive blood, and King says that raises questions about the ethics of the study's design.
"In this trial, if somebody comes to the hospital and they've been randomized to get PolyHeme in the field, they're going to keep getting PolyHeme for up to 12 hours after they arrive at the hospital. And that's at least 11 hours after they could have received typed and cross-matched whole blood, which is the standard of care.
"And since the regulations themselves say that it's only appropriate to use this emergency waiver of consent when available treatments are unproven or unsatisfactory, once they get to the hospital, and typed and cross-matched whole blood is available, the trial no longer meets the condition for meeting the emergency waiver."
Why then would the Institutional Review Board at UCSD buy into such a study?
"I think there are two issues going on," says King. "One is that there is not a lot of activity under the waiver, and there are a lot of IRBs who are very, very busy and understaffed and all that kind of stuff, who don't know the regulations well. They may actually look at a trial like this and say, 'Well, the FDA is really the expert in these regulations, and if the FDA says it's okay, it must be okay.' "
King says she has reservations about the City of San Diego's decision to test PolyHeme only in largely poor and minority neighborhoods. "I think that the kind of targeting that you're describing, if that were known, that would cause quite a stink.
"I think it's an issue with a blood-substitute study where probably the two populations most likely to be enrolled are people who have been in motor vehicle accidents and people who have experienced violence.
"The assumption that particular neighborhoods are going to produce more subjects or the assumption that particular neighborhoods are going to produce subjects whose families are less likely to ask questions afterwards is problematic."
In a brief telephone interview he granted Monday, Hoyt defended UCSD's participation in the PolyHeme study against critics such as King. "The exception to informed consent is a policy that is fully legitimate and a process that has been recognized and approved by the federal government," Hoyt said, adding that the protocol is not only legitimate and safe, it is appropriate and a way to extend important medical research. He added, "We have very broad support throughout the community."
The issue of notifying residents of neighborhoods in which the PolyHeme-equipped rigs are located "wasn't something we specifically considered" as part of the study's outreach effort, he said.
Neighborhoods in the city of San Diego where PolyHeme is deployed were chosen based on their trauma rates. There was a "cost containment problem," as described in the Romeril memo, because it would have been too costly to deploy the PolyHeme on every paramedic unit, he said.
Regarding the adverse event reports, he said that if a patient had a serious illness or died and it wasn't due to the PolyHeme, that would be an example of an adverse event. As to who determines whether the substance was responsible or not, that is a "consensus" decision made by him and other research staff, and reported to the Institutional Review Board. No adverse events related to PolyHeme had occurred, Hoyt added.


It reads like a bad science fiction novel: A small Illinois biotech company cuts a deal with UCSD. The university agrees to test a substitute for human blood on comatose patients — victims of gunshots and car crashes — without the patients' consent. Within the city of San Diego, the experiment is targeted at several neighborhoods south of I-8, where many poor and minority residents are unlikely to have heard of the study and unlikelier still to have the resources to sue if something goes awry. The university conceals the identity of the city's paramedic units who carry the blood substitute.

When a curious reporter asks for the names of the neighborhoods where the study is being carried out, a research coordinator working for the university tells him that the Chicago sponsor doesn't want the information made public because Wall Street moneymen — hoping to reap a financial windfall from their investment in the company — might sell if they discover the experiment is not going well. When the reporter asks university officials to provide details of two "adverse event" reports involving the study, the officials stonewall, saying the information is "nonpublic" because the university has signed a confidentiality agreement with the laboratory. But if Hollywood wouldn't buy that scenario, the University of California and the City of San Diego already have. A little more than two years ago, officials at UCSD signed an agreement with Northfield Laboratories for clinical trials of PolyHeme.

Despite repeated studies, PolyHeme has yet to be approved by the Food and Drug Administration, and this May, Forbes magazine told its predominantly Wall Street audience that researchers at Walter Reed Army Institute of Research had "published a study suggesting PolyHeme might not be so safe after all. Comparing resuscitative fluids, it found that hemorrhaging lab rats treated with PolyHeme experienced higher mortality rates than those that received two commonly used solutions."

First, hemoglobin is extracted from red blood cells and filtered to remove impurities. This purified hemoglobin is then chemically modified and purifies using a multi-step process to create a polymerized form of hemoglobin. The modified hemoglobin is then incorporated into an electrolyte solution.
Polymerization is intended to eliminate the undesirable effects historically associated with hemoglobin-based blood substitutes: vasoconstriction, kidney dysfunction, liver dysfunction, and gastrointestinal distress.
The founder of Northfield labs is Steven Gould, a 57-year-old physician from Chicago who has worked on blood substitutes for more than 25 years. In 1979, after obtaining a grant from the Army, he and several other doctors at the University of Chicago developed a technique to separate hemoglobin, the oxygen-carrying protein, from blood, then link the hemoglobin molecules together into polymers, hence the name PolyHeme.
"Initially a project of the United States Army following the Vietnam War," Northfield's website says, "the objective was to develop a preparation that could be infused rapidly and in massive quantities to injured battlefield combatants in far-forward areas and mobile surgical theaters before the injured could be evacuated to more sophisticated field hospitals for definitive care."
Northfield's promotional material describes PolyHeme as "a solution of chemically modified human hemoglobin, which simultaneously restores lost blood volume and hemoglobin levels and can be rapidly and massively infused." The advantages of PolyHeme are that it "requires no cross-matching. It is compatible with all blood types and therefore immediately available for infusion. It has an extended shelf life of over 12 months."
The Army continued to fund Gould until 1984, when it began supporting his larger rival, Baxter International, in its own quest for a blood substitute. At that point, Gould shifted his aim and put together $1 million in funding from private investors. A decade later, in 1994, with several small clinical trials under his belt, Gould took Northfield public, raising $15 million at $6.50 a share.
But in 2001, according to the Forbes article, the Food and Drug Administration rejected Northfield's application to approve PolyHeme for general use, citing lack of sufficient clinical studies. The stock tanked, sinking from $13.25 to $3.05.
Gould held on, sacking the company's chief executive and eventually reaching an agreement in 2003 with the FDA to allow a new kind of clinical trial that could be run without getting the patient's consent. Employing a little-used exemption in FDA regulations, Gould proposed having paramedics in cities across America infuse PolyHeme into 360 patients. San Diego was one of those cities.
Under Northfield's deal with the FDA, the company and its research partners agreed to conduct public information campaigns in each city where PolyHeme was to be deployed. They were to arrange newspaper, radio, and television coverage advising the public of the tests and of the opportunity to opt out by wearing a special bracelet. They were to set up a website containing voluminous information about the nature of the study and the right to opt out. In San Diego, much of this didn't come to pass.
Notice of the trial was posted on an obscure part of a website run by the UCSD Human Research Protections Program. Today, those who click on a link labeled "Public Notices" are taken to another link labeled "PolyHeme Trauma Trial" and thence to the study's information page. Readers are told that "This groundbreaking study is being conducted in accordance with federal regulations permitting an exception from informed consent requirements under 21 CFR 50.24." The statement says that a public information meeting, where "comments and concerns" were to be solicited, would be held August 6, 2003, at the UCSD Medical Center in Hillcrest. "Members of the media are invited to cover this event," it added.
The only major newspaper story covering the trial was spun heavily in favor of the wonders of the experimental substance and made light of its possible hazards. "A new hope for saving of lives," said the headline over the March 23, 2004, story in the Union-Tribune. "Paramedics to conduct test of blood substitute."
"These are people who are facing death in a few hours," the story quoted Dr. David Hoyt, trauma director at UCSD Medical Center and principal investigator of the local PolyHeme study. "Giving them this blood substitute might just save them when conventional treatment wouldn't." The paper added that "In a study of PolyHeme from the late 1990s, 75 percent of those treated with it survived, compared with 30 percent treated only with saline solution."
The paper went on to report that "the blood substitute will only be available to paramedics working south of Interstate 8." Specific paramedic and ambulance units carrying PolyHeme were not identified. "Designated paramedic units in San Diego and the South County and the Mercy Air helicopter crew will have a cooler containing bags of the substitute," the story added.
The clinical trial began in San Diego County on May 20, 2004, when a Mercy Air emergency-services helicopter, based at Gillespie Field, started carrying PolyHeme. On June 11, 2004, seven ground ambulances serving cities outside of the city of San Diego joined the trial. The ground ambulances are owned by a private company, American Medical Response, which serves 11 cities in the county as well as unincorporated areas. According to the company's spokesman, Devon Price, the ambulances participating in the study operate in National City, Chula Vista, Bonita, and Imperial Beach. Earlier this week, Hoyt, of UCSD, said six paramedic rigs outside the city of San Diego were equipped with PolyHeme.
But according to records obtained from the City of San Diego, at the time the U-T story appeared, city officials and UCSD administrators were still negotiating when and where the city's paramedics would use PolyHeme.
Northfield wanted to limit the PolyHeme trial to neighborhoods with the highest trauma rates. On May 2, 2003, Beth Romeril, the research coordinator of the UCSD study, wrote Hoyt, the principal investigator. "My biggest concern at this time is that in conversation with Bob McGinnis from Northfield on Wednesday, he indicated to me that Northfield would not have enough product to stock even the 12 or so ambulances in UCSD's catchment area, that part of the reason for the TREG [San Diego County Regional Trauma System Registry] search would be to identify the '3 or 4' ambulances that would be most likely to encounter appropriate patients.
"I have asked him to confirm the maximum number of ambulances they would be able to supply for us and waiting to hear back," Romeril wrote, adding that her geographic survey of trauma data was complicated. "Most of the time the location (zip code) where the encounter took place is not available, so we can't identify specific medic rigs in most cases."
At the City of San Diego, officials worried about possible lawsuits in case PolyHeme didn't work as advertised. They wanted to isolate the city's possible financial liability by demanding that the study's sponsor indemnify the city for $15 million against malpractice claims.
Why might there be malpractice claims?
By the very definition of the study, all patients are in hemorrhagic shock and unable to refuse the PolyHeme. As noted by Northfield: "It is expected that patients enrolled in this trial will be unable to provide informed consent because of the nature and extent of their injuries." The lack of consent was justified on the grounds that the existing blood substitute currently used by paramedics is simple saline solution and that almost anything, even an experimental substance like PolyHeme, would be an improvement for desperate trauma patients.
And there was another reason for the city to fear lawsuits. Even after arriving at the hospital, patients would not receive real blood.
The protocol called for trauma patients selected for the study to be randomly separated into two groups: the control group would get the standard saline as the resuscitative fluid; the experimental group would be infused with PolyHeme. When patients arrived at the hospital, those receiving saline would be switched to blood. But those receiving PolyHeme would continue to receive PolyHeme -- up to a maximum of six units -- for 12 hours after their injury.
Other medical centers were not as sanguine as UCSD about the PolyHeme trial and pulled out before it began. According to a letter from Northfield to Hoyt, dated last August 27, Milwaukee's Froedtert Memorial Lutheran Hospital's review board "has not approved conduct of the study at their institution. The letter from [redacted] cites this [review board's] position that once the patient is in the hospital, blood is not an unsatisfactory treatment, which is what is required for waived consent."
Boston Medical Center dropped out for similar reasons.
A study at the University of Southern California was shut down before it began, according to state records.
The City of San Diego moved forward with the trial in midsummer. In a memo dated August 10, 2004, the city's Emergency Medical Program manager, Donna Goldsmith, quietly informed the council that the PolyHeme trial would finally proceed. "Based upon previous trauma demographic data," the memo said, "it is projected that the San Diego region will contribute 40 patients to the study. The safety and efficacy of this artificial blood product have been demonstrated in several in-hospital studies involving more than 300 patients."
Goldsmith's memo went on to say that paramedic units in three of the city's poorest communities -- Oak Park, Nestor, and San Ysidro -- had been chosen for the trial because those areas generate the largest number of severe trauma patients, who typically are victims of car accidents and shootings. (A fourth paramedic unit, based downtown, would later be included in the study, Goldsmith said in a recent telephone interview, but that was not included in the memo to the council.)
"Specific ambulances and medic-engine crews have been selected to carry PolyHeme in specially designed coolers," the memo said. "The crews selected are those that frequently encounter major trauma victims within the catchment regions of the trauma centers participating in the trial -- UCSD Medical Center and Scripps-Mercy Hospital."
The units assigned to the PolyHeme trial -- Oak Park's Medic 26, San Ysidro's Medic 29, and Nestor's Medic 62 -- rarely venture from their home turf, Goldsmith said.
"Although PolyHeme will be placed on the engines and ambulances listed above, due to the fluidity of the EMS System, these resources could be assigned as needed throughout the City."
But under the terms of the study, paramedics could administer PolyHeme only if they were delivering the patient to the UCSD Medical Center or to Scripps Mercy Hospital.
The agreement did not go to council. It is dated September 28, 2004, and is signed by then-deputy city manager Patricia Frazier. The trial began in the city on November 19.
(In May of this year, Frazier, along with former treasurer Mary Vattimo and human resources director Cathy Lexin, quit her job in what was widely seen as a shake-up related to the city's festering pension-fund crisis. Lexin and Vattimo, former members of the pension board, were later charged with conflict of interest for their votes to allow underfunding of the pension system. Frazier was not charged.)
Though they had become part of the nationwide PolyHeme trials, residents of the neighborhoods served by the designated paramedic units were none the wiser. Many details of the PolyHeme study's protocol, along with much other information about the trial in San Diego, have never been made public. In a letter dated January 16, 2004, Gould told Hoyt, principal investigator of the UCSD study, "I urge you to remind your study coordinator and all others associated with the trial that information about patients and the details of the trial at your site are not to be shared with anyone outside the study team."
Under its August 2004 agreement with UCSD, the City of San Diego agreed "not to disclose to any third party any Confidential information," defined as "all information, data, materials, in whatever form or medium, that [Northfield] treats as confidential and proprietary information."
The secrecy has been sweeping, denying citizens any chance to monitor the study's progress or receive information about possible adverse effects on patients. A "research plan," dated January 10, 2003, and attached to the city's contract with UCSD, was obtained from the city after repeated requests under the California Public Records Act. The plan spells out details of the PolyHeme trial, but before UCSD turned it over earlier this year, it was heavily redacted to remove any hint of where and how the substance is being tested or what paramedics are to do in case of life-threatening side effects.
One section of the plan, which describes how UCSD researchers would attempt to get the consent of a comatose patient's relative before proceeding, was gutted. It begins:
"Once a patient is enrolled, the investigator will commit to diligent and frequent attempts to contact a legally authorized representative or a family member in order to provide them with the opportunity to object to the patient's continued participation in the study. At a minimum, contact with a legally authorized representative or family member by the study coordinator will be attempted at least:"
The paragraph that follows, apparently describing the mandated conditions for obtaining consent before proceeding, is completely blacked out.
Particularly troubling to some observers are two in-house university reports of "adverse events" that have occurred.
A letter dated May 27, 2004, from Daniel Masys, M.D., then director of UCSD's Human Research Protections Program, to Hoyt discussed the first incident. "Your April 29, 2004 adverse event report for Project 030443 has been reviewed and accepted by the IRB Committee at the May 20, 2004 meeting," Masys wrote. The IRB, or Institutional Review Board, is a group set up by the university to review the ethics and integrity of research activities.
The letter's next sentence, apparently describing the nature of the adverse event, was entirely redacted by the university. Two final sentences followed the redaction: "The UCSD IRB believes that all issues have been adequately addressed and feels that the decision to support this protocol in July 2003 was appropriate. Thank you for keeping us informed."
Asked how an adverse event could have occurred in April 2004, almost a month before the study began on May 20, Beth Romeril, the UCSD research coordinator affiliated with the study, denied that the May 27, 2004, letter regarding an adverse event report had anything to do with the PolyHeme study, even though the document specifically refers to the trial. Romeril added that university staff fill out a lot of forms and sometimes forget to change the heading at the top, so they might have left the wrong project number on the document. She offered no explanation as to why the letter also makes reference to the project number in its body.
Another letter from Masys to Hoyt, dated August 26, 2004, referenced a subsequent incident. "Your July 22, 2004 adverse event report for Project 030443 has been reviewed and accepted by the IRB Committee at the August 2004 meeting. This report concerns the following subject: 1)." An entirely redacted sentence, apparently describing the specifics of the adverse event, followed.
The letter then concluded: "It is your opinion that the event is definitely unrelated to the study. It is also your opinion that this update will not require a change to the risk management procedures, application, informed consent document, or the risk/benefit ratio. Thank you for keeping us informed."
When asked to provide copies of the reports referred to in the letters, the university produced a form labeled "Report of Adverse Event," signed by Hoyt, dated July 27, 2004. Two pages of the form, under the heading "Principal Investigator's Assessment of Event," were completely blank, with the exception of the word "redacted" stamped repeatedly down the empty pages. Other information, such as the date of occurrence and whether it caused a fatality or was life-threatening, was also blacked out.
In the April 28, 2005, cover letter that accompanied the material, Linda L. Maczko, the university's Public Records Act coordinator, said, "The basis for the redaction to the adverse event report and consent form is patient privacy.... The basis for all the other redactions made to the documents provided is trade secret/proprietary information."
Legal experts familiar with public records law note that patient privacy can be protected by redacting the patient's name and identifying information and thus is not grounds to withhold other details of the event.
They also maintain that in such situations where a public agency may place members of the public at risk without their consent, the state's Public Records Act requires the agency to provide the public with as much information as possible.
"We filed this records request originally with UCSD on December 23, 2004," notes William Sauer, the attorney who has handled the Reader's repeated Public Records Act requests to the university. "UCSD has continually 'shielded' the results of the trial, alleging it is a trade secret and its release violates patient confidentiality.
"We have asked for no individual patient information. We have asked for no secret formulas or processes. UCSD is a public university that should respond promptly to the CPRA. Instead it shields and protects drug companies that pay it for the clinical trials.
"UCSD should carefully review the recent overwhelming approval by the voters in California of Prop. 59, commonly referred to as the Sunshine Amendment. Frankly, we haven't seen delaying tactics like this in 25 years of legally obtaining public documents."
Under a "clinical study agreement" between UCSD and Northfield dated March 22 of last year, the university is to collect $10,840 "per Completed Patient" in the study. Forty patients were anticipated.
The contract itself -- provided by the university, but heavily redacted -- outlines a "payment schedule" based on how long a patient remains in the trial. If a patient drops out early in the course of treatment, before the goals of the timeline are met, UCSD receives significantly less money. That might create a conflict of interest, says Nancy King, a bioethicist, between the university's ethical mandate to fully inform patients and relatives about the dangers of the treatment and the university's desire to get fully paid by Northfield.
Because the contract was heavily redacted, a full understanding of the payment schedule is difficult. For example, the contract says that "for each Qualified Patient completing step 1 who withdraws from the study for any reason prior to completing [redacted]," Northfield will pay the university just 35 percent of the $10,840 fee. "For each Qualified Patient completing step 1 [redacted] phase," the fee would be 50 percent of the total, and so forth.
No matter how many patients are enrolled or how long they remain in the study, the contract says, "Non-refundable Start Up costs of $39,106.00 plus the cost of four (4) completed patients ($43,360.00 less 20% ($8,672) = $34,688) for a total of $73,794.00 will be payable upon full execution of the Agreement." All costs are subject to a 3 percent annual adjustment for inflation.
Scripps Mercy Hospital is also paid for participating in the study. According to a contract between UCSD and Scripps Health, dated May 11 of last year, UCSD sought to "conduct part of this trial" at Scripps Mercy "in order to enroll a larger number of patients." Using funds provided by Northfield, the contract says, UCSD agreed to pay Scripps Mercy Hospital a $2000 fee for each trauma patient who completed the PolyHeme trial, as set out in its agreement with Northfield. Of that $2000, the hospital receives $878 for lab fees and Dr. Michael Sise, head of trauma care at Scripps Mercy, collects $1122 per patient, the contract says.
According to an e-mail from Scripps Health public relations director Don Stanziano, the payment to Sise is not for his sole benefit. "While reimbursement is sent to Dr. Sise -- lead investigator at Scripps Mercy, he (the Scripps Mercy Trauma Department) distributes the monies to cover the costs as outlined in the study's approved budget.
"Physicians receive reimbursement for the extra time it takes to attend to the study. The trauma surgeons (four at Scripps Mercy) receive $560 per patient. [That] money is donated to Scripps Mercy's research fund to offset the cost of doing unfunded clinical research. The remaining funds pay for other staffing costs such as nursing."
Stanziano declined to comment on whether Mercy had suffered any adverse events related to the study. "Due to confidentiality of study protocols, that question must be directed to Northfield Labs, the study's sponsor," the e-mail said.
Under state law, the medical director of the county's Emergency Medical Services, Dr. Gary Vilke, is supposed to act as watchdog over all paramedics in San Diego County. But that is not Vilke's only job. The rest of his time is spent as medical director of prehospital services at UCSD Medical Center, where Hoyt is a member of the board of governors.
In the fall of 2003, Vilke sought permission from Richard E. Watson, the interim director of the state's Emergency Medical Services Authority, to begin testing PolyHeme in San Diego. "Initially I had concerns about the study due to the exception for patient consent," Watson responded to Vilke on October 23, 2003.
"My approval, however, is based on your agreement to do the following. For the first two months following initiation of the trial study, I would like a weekly update on the study via a phone call to me or Dr. Tharratt. For the duration of the study would also require immediate notification (by phone or fax) to Dr. Tharratt of any occurrence of a serious adverse event...and a written trial study progress report every 6 months."
Vilke reported to Watson on the study's progress in a letter dated last December 16. Despite the two adverse event reports on file at UCSD, Vilke reported that "There were no complications in the study protocol, nor were there any complications or adverse events in the care of the patients in pre-hospital or hospital setting."
Dr. Steven Tharratt, who is a medical consultant to the state's Emergency Medical Services Authority, says the authority has not been provided copies of the two adverse event reports filed by Hoyt. Despite Watson's requirement that Tharratt be notified immediately of a serious adverse event, Tharratt said the adverse event reports sent to the IRB "wouldn't be something we would be interested in seeing. That is not our area of oversight."
Vilke also reported in his letter to Tharratt that as of last December, the UCSD study had enrolled only four patients. Vilke added, "All 4 patients enrolled in the study are doing well."
Six months later, after he had been queried by a reporter about the adverse event reports, Vilke reported to Watson, in a letter dated June 20, 2005, that "to date, we have enrolled a total of fourteen patients into the study.
"There have been a total of four protocol violations. All have been reviewed by me and none resulted in any adverse events with the patients. There were no complications as a result of the study protocol, nor were there any complications or adverse events in the care of the patients in pre-hospital setting.
"We have not had any immediately reportable safety events, so no IND safety reports have been generated to date. All patients enrolled have done well and are all alive at 30-day follow up.
"We have monitored the scene times for these patients and they were essentially unchanged compared with similar status trauma patients during the same time period."
According to Vilke's letter, at least 250 patients from across the nation have been enrolled so far in the study.
But finding suitable test subjects has not been easy. Struggling to recruit subjects to the PolyHeme trials, researchers in December considered offering paramedics "incentives" in return for their cooperation, according to documents released by UCSD after more than five months of delay.
"Are there any incentives/activities that would motivate the EMS that you use or would recommend using that would not be inappropriate or be equated to directly rewarding them for patient recruitment?" asks Northfield in an agenda, dated December 8, 2004, for an "Enrollment Meeting" at its Evanston, Illinois, headquarters. The apparent response from UCSD, highlighted in boldface type: "Gifts of food items (Christmas baskets: coffee) or inexpensive give-aways like pens, penlights, mugs with 'Polyheme study' on them for the whole group regardless of enrollment as a thank-you for participation."
The agenda asks, "Do you know of any incentive program restrictions that might exist at your hospital?" The response: "No monetary gifts or payment for enrollment."
The document also said, "Share with us the worst comment you've heard from EMS/Flight Nurses (or any other study staff) regarding the study. What would be a creative response?"
The reply: "Flight crew from North County: We bring you maybe one trauma patient/year that would qualify for this study, so why did they bother including us?
"We have responded that there are so few patients that meet criteria, that every single one is precious for the study, plus the fact that the air crews are potentially the most reliable at following study protocols and thus providing the best data."
One of the most outspoken critics of the PolyHeme study is Nancy King, a professor of social medicine at the University of North Carolina at Chapel Hill School of Medicine. Trained as a lawyer and specializing in bioethics and health law, King has concentrated on issues of informed consent, a specialty that ultimately led her to monitor closely the genesis of the PolyHeme study.
"When one gets involved in a research study," King says, "normally one goes through a fairly extensive consent process and signs a consent form and is given a lot more information about what's going to happen than may well happen in the doctor's office when the doctor says, 'I think you need such and such.' "
That started to change back in 1996, King says, when Baxter International, the large pharmaceutical company, petitioned the federal government to allow testing of its blood substitute on patients who could not provide their consent.
"That's a very big departure from all of the regulatory structures that govern human-subjects research. What was ultimately approved was a regulation that permitted research under those limited circumstances where the subject is in a life-threatening condition.
"One of the things that has been quite interesting to follow since that regulatory structure was put in place was...well, there haven't been that many studies done under the emergency waiver. And people have a lot of questions about how to do it well."
Is the waiver of informed consent necessary to conduct medical research? King says there may be circumstances that make it impossible to require patient consent before an experimental treatment is administered, but they are few and far between.
"I think that most of the time, and frankly almost all of the time, there are ways to do relevant studies where the waiver is not needed. What the waiver gets you really is faster enrollment of subjects.
"You may have a scientific reason for saying we need to get more subjects quickly, but more often it's a question of how fast a drug or device or procedure that's new needs to move through the system in order to be most profitable for the study sponsor.
"One of the concerns that I have about the PolyHeme trial is that, looking at the study sponsor's website, their materials basically talk about the future of PolyHeme as being in military applications and for elective surgery. In both of those populations one can get prospective consent. So the very design of the PolyHeme study is troubling since there are other ways to gather the data.
"The other thing that has troubled me -- and this, I think, is a flaw in the IRB process as well as in the way the sponsor's thinking about the study -- is that the kinds of things that are being done per community consultation and the material that's available for community consultation are very slim.
"Most sites have merely posted notices about public meetings held in places that people don't usually go, and the investigator will go and give the slide show that's given to him by the study sponsor and hand out materials that have been developed by the study sponsor, which is all the same stuff [at every site].
"And it's all fairly flawed, because the basic materials, the basic FAQ sheets about the study, talk about how many 'patients' have already been 'treated' with PolyHeme, and that's a very misleading description.
"Nobody who has gotten PolyHeme has been anything but a research subject, because this is an experimental intervention. So anybody who gets it is a research subject. And it's not a treatment yet because it hasn't been approved. So saying, 'Well, X number of patients have already been treated with PolyHeme' provides a false sense of reassurance to a subject that may actually be quite misleading."
Northfield has carefully guarded its research protocol, the plan detailing how its study is to be conducted, the dosage to be used, the results of prior testing, and other details. King argues that this is common in the drug industry but not appropriate for research on subjects who do not provide their consent.
"What was shown in earlier-phase research? What kind of earlier-phase research was actually conducted? What numbers are there? What leads up to saying this is the right time for this kind of study now?"
Without such information, King says, communities can't judge the trade-offs between PolyHeme and saline solution, the currently used alternative.
"What ambulances can do is give you saline to keep up your blood volume, but they can't actually provide you with blood that is able to enable your system to keep getting enough oxygen," she explains. "They've got to give you saline and try to stop the bleeding and get you to the hospital, where you can get blood, because that's what carries oxygen.
"In the field then, if the argument is that PolyHeme can supply some of that oxygen at least temporarily, and it doesn't need to be refrigerated, and it doesn't need to be typed and cross-matched, well, that would be a really good thing to have."
But once a patient gets to the hospital, where blood is available for transfusion, the equation changes. PolyHeme-study patients are forbidden to immediately receive blood, and King says that raises questions about the ethics of the study's design.
"In this trial, if somebody comes to the hospital and they've been randomized to get PolyHeme in the field, they're going to keep getting PolyHeme for up to 12 hours after they arrive at the hospital. And that's at least 11 hours after they could have received typed and cross-matched whole blood, which is the standard of care.
"And since the regulations themselves say that it's only appropriate to use this emergency waiver of consent when available treatments are unproven or unsatisfactory, once they get to the hospital, and typed and cross-matched whole blood is available, the trial no longer meets the condition for meeting the emergency waiver."
Why then would the Institutional Review Board at UCSD buy into such a study?
"I think there are two issues going on," says King. "One is that there is not a lot of activity under the waiver, and there are a lot of IRBs who are very, very busy and understaffed and all that kind of stuff, who don't know the regulations well. They may actually look at a trial like this and say, 'Well, the FDA is really the expert in these regulations, and if the FDA says it's okay, it must be okay.' "
King says she has reservations about the City of San Diego's decision to test PolyHeme only in largely poor and minority neighborhoods. "I think that the kind of targeting that you're describing, if that were known, that would cause quite a stink.
"I think it's an issue with a blood-substitute study where probably the two populations most likely to be enrolled are people who have been in motor vehicle accidents and people who have experienced violence.
"The assumption that particular neighborhoods are going to produce more subjects or the assumption that particular neighborhoods are going to produce subjects whose families are less likely to ask questions afterwards is problematic."
In a brief telephone interview he granted Monday, Hoyt defended UCSD's participation in the PolyHeme study against critics such as King. "The exception to informed consent is a policy that is fully legitimate and a process that has been recognized and approved by the federal government," Hoyt said, adding that the protocol is not only legitimate and safe, it is appropriate and a way to extend important medical research. He added, "We have very broad support throughout the community."
The issue of notifying residents of neighborhoods in which the PolyHeme-equipped rigs are located "wasn't something we specifically considered" as part of the study's outreach effort, he said.
Neighborhoods in the city of San Diego where PolyHeme is deployed were chosen based on their trauma rates. There was a "cost containment problem," as described in the Romeril memo, because it would have been too costly to deploy the PolyHeme on every paramedic unit, he said.
Regarding the adverse event reports, he said that if a patient had a serious illness or died and it wasn't due to the PolyHeme, that would be an example of an adverse event. As to who determines whether the substance was responsible or not, that is a "consensus" decision made by him and other research staff, and reported to the Institutional Review Board. No adverse events related to PolyHeme had occurred, Hoyt added.
Comments